Parasitism is an unequal relationship, where one species benefits at the expense of a host species which is negatively affected in some way. This interaction can diminish host survival and reproductive capabilities, and even lead to the host’s death. Some researchers suggest that differences between parasitism and other symbiotic relationships, like mutualism, are not so large and strict; they could shift in response to environmental changes (1).
In a very general classification, there are two major groups, ectoparasites and endoparasites. The main difference between those groups is the location of the parasite: ectoparasites live on the body surface of the host and endoparasites inhabit their guts or cavities. The negative effects of parasites can vary from strictly physical to immunological or chemical.
Typical endoparasites are Acanthocephala (thorny-headed worms) or Cestoda (tape worms) which infest fish and other animals and feed on the nutrients in the intestines of their hosts. Other endoparasites may affect their hosts by excretion of toxic substances as a result of their metabolism (2).
Another example of endoparasite is the microsporidian fungus Glugea. This genus is one of the largest among the microsporidian fungi, with at least 1300 to 1500 species. They are intracellular parasites in a wide range of hosts, including commercial fish species, and tend to grow single-cell tumours (3). Microsporidiosis infection in aquaculture and wild fish is well known by scientists and has a considerable economic impact in the fisheries sector (4).

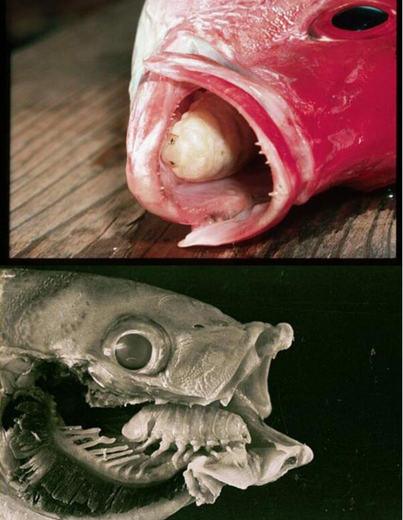
The isopod shrimp Cymothoa exigua, another well-known fish parasite, has a rather interesting relationship with its host. The larvae of this species enter through the gills of the fish. Only the females attach their legs to the tongue, cutting off some of the blood supply. The tongue deteriorates usually only in part and the isopod feeds on the food entering the fish’s mouth, but also on the host’s fluids. The parasite seems not to do any visible harm, but some studies suggest that the presence of Cymothoa exigua probably leads to an earlier death, especially if they enter during the first years of the fish’s life (5, 6).
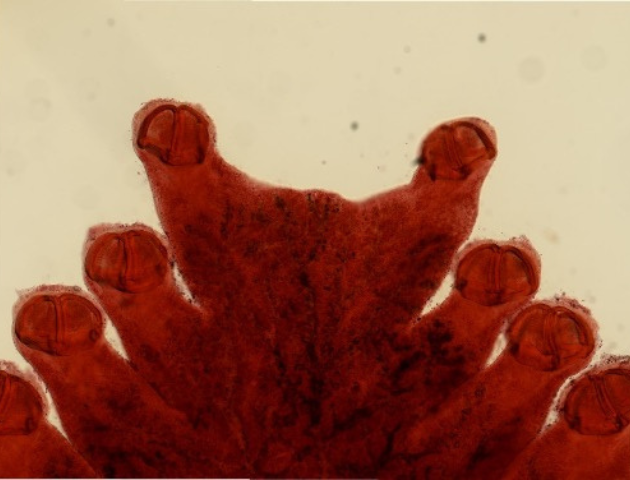
Parasites being part of every ecosystem, a number of studies have also focussed on the effects of pollution, climate change and species invasion on these groups of organisms. They have been used widely as indicators of eutrophication (accumulation of nutrients) or for detecting hydrocarbon and heavy metals in the environment and are considered as valuable as free-living animals to gather information about pollution (7). Diclidophora merlangi, a marine flatworm, infesting the gills of the whiting Merlangius merlangus, is a very valued bioindicator of hydrocarbon pollution and has a long history of use in the North Sea. A study published in 2022 revealed a significant increase in numbers of infested fish the nearer they are to an oil field (8).
The effects of climate change and global warming on parasites, especially in marine environments, are not yet fully understood. More research is needed to better predict the response of parasitic organisms to a changing climate, but some studies suggest that for those parasites not killed by the increasing environmental temperatures, intensity and pathogenicity might increase (9). In other words, as sea temperatures rise, some parasites will die, but others will thrive and cause more harm to their host population.
Author: Dr Loreto Gestoso Suarez
References
1. Leung T. L. F. & Poulin R. (2008). Parasitism, commensalism, and mutualism: exploring the many shades of symbioses. Vie et Milieu – Life & Environment, 58 (2): 107-115.
2. Rohde K. (1984). Ecology of marine parasites. Helgoländer Meeresuntersuchungen: 37: 5-33.
3. Abdel-Baki A.-A.S., Al-Quraishy S., Rocha S., Dkhil M.A., Casal G., Azevedo C. (2015). Ultrastructure and phylogeny of Glugea nagelia sp. n. (Microsporidia: Glugeidae), infecting the intestinal wall of the yellowfin hind, Cephalopholis hemistiktos (Actinopterygii: Serranidae), from the Red Sea. Folia Parasitologica, 62: 007. doi: 10.14411/fp.2015.007
4. Mansour L., Thabet A., Harrath A.H., Al Omar S.Y., Mukhtar A., Sayed S.R. & Abdel-Baki A-A. Sh. (2016). New Microsporidia, Glugea sardinellensis n. sp. (Microsporea, Glugeida) found in Sardinella aurita Valenciennes, 1847, collected off Tunisian coasts. Acta Protozoolica, 55: 281-290.
5. Chávez Dagostino R., Bravo M. & Gómez Morales N. (2016). Cymothoa exigua Schioedtes & Meinert, 1884 (Isopoda: Cymothoidae) en el Pacifico Este. Acta pesquera, 4: 42-48.
6. Ruiz-L A., & Madrid-VJ. (1992). Studies on the biology of the parasitic isopod Cymothoa exigua Schioedte and Meinert, 1884 and its relationship with the snapper Lutjanus peru (Pisces: Lutjanidae) Nichols and Murphy, 1922, from commercial catch in Michoacan. Ciencias Marinas, 18(1): 19-34.
7. Williams H & Mackenzie K. (2003). Marine parasites as pollution indicators: An update. Parasitology, 126 Suppl. (7): 27-41.
8. Sue H., Mackenzie K., Ives S., Pert C. & Jones C. (2022). Diclidophora merlangi (Kuhn, 1829) Krøyer, 1838 (Monogenea: Diclidophoridae) as an indicator of hydrocarbon pollution in the North Sea. Marine Pollution Bulletin, 185, part A: 114268. doi:10.1016/j.marpolbul.2022.114268
9. Byers J. (2021). Marine Parasites and Disease in the Era of Global Climate Change. Annual Review of Marine Science, 13: 397–420.
Photographic credits
Figure 1: Hans Hillewaert https://commons.wikimedia.org/wiki/File:Glugea_stephani.jpg
https://creativecommons.org/licenses/by-sa/3.0/deed.en
Figure 2: Matthew R. Gilligan https://www.flickr.com/photos/oceanexplorergov/50856153641/in/photostream/
https://creativecommons.org/licenses/by-sa/2.0/
Figure 3: Daniel J. Drew https://commons.wikimedia.org/wiki/Category:Diclidophora_merlangi#/media/File:Diclidophora_merlangi_(YPM_IZ_097932).jpeg
https://creativecommons.org/publicdomain/zero/1.0/deed.en











